Nuclear Fission
Understand how splitting heavy nuclei releases enormous energy
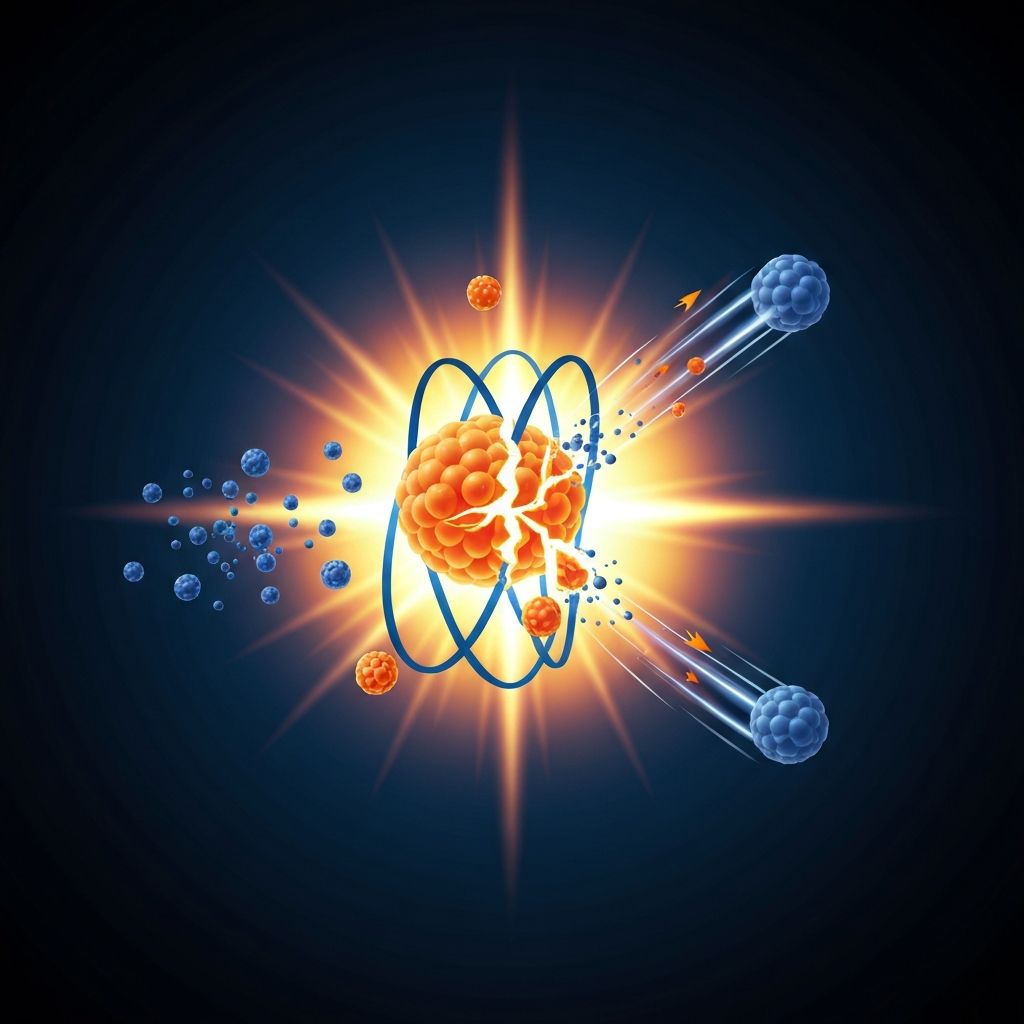
Splitting the Atom
Releasing the power within the nucleus
Nuclear fission is the splitting of a large, unstable atomic nucleus into two smaller daughter nuclei. The most common fissile materials are uranium-235 and plutonium-239, used in nuclear reactors and weapons.
Fission can occur in two ways: spontaneous fission (rare, nucleus splits without external trigger) and induced fission (a neutron is absorbed, making the nucleus unstable and causing it to split).
When a neutron hits a uranium-235 nucleus, the nucleus absorbs it and becomes uranium-236, which is highly unstable. This unstable nucleus immediately splits into two smaller daughter nuclei (such as barium-144 and krypton-89), releasing 2-3 neutrons and gamma rays.
235U + 1n → 144Ba + 89Kr + 31n + Energy
Each fission event releases approximately 200 MeV (200 million electron volts) of energy—mostly as kinetic energy of the products, plus heat and gamma radiation.
The total mass of the fission products is slightly less than the original uranium nucleus plus neutron. This "missing" mass is called the mass defect. According to Einstein's famous equation, this mass is converted directly into energy:
E = mc²
Even a tiny mass defect produces enormous energy because c² (speed of light squared) is so large
The 2-3 neutrons released from each fission can trigger more fissions in neighboring uranium nuclei. This creates a chain reaction with exponential growth: 1 → 2 → 4 → 8 → 16 → 32...
- Controlled chain reaction: In nuclear reactors, control rods absorb excess neutrons to maintain a steady reaction rate for power generation.
- Uncontrolled chain reaction: In nuclear weapons, the chain reaction is allowed to proceed unchecked, releasing massive energy in microseconds.
The minimum amount of fissile material needed to sustain a chain reaction is called the critical mass.
Fission Equation:
235U + 1n → 144Ba + 89Kr + 31n + Energy
Energy released: ~200 MeV per fission event
Mass-Energy Conversion (E = mc²)
The products have slightly less mass than the original uranium + neutron. This "missing mass" (mass defect) is converted to energy according to Einstein's equation.
Mass defect: ~0.2 atomic mass units → ~200 MeV of energy
Nuclear Fission
Question:
Complete the fission equation: 235U + 1n → 141Ba + ? + 31n
Answer:
Step 1: Balance mass numbers (top numbers)
Left side: 235 + 1 = 236
Right side: 141 + ? + 3(1) = 141 + ? + 3
So: ? = 236 - 141 - 3 = 92
Step 2: Balance atomic numbers (bottom numbers)
Uranium = 92, Barium = 56
92 + 0 = 56 + ? + 0, so ? = 36 (Krypton)
Answer: 92Kr (Krypton-92)
What is nuclear fission?