Alkenes – Structure, Reactions, and Tests
Understanding unsaturated hydrocarbons and their characteristic reactions
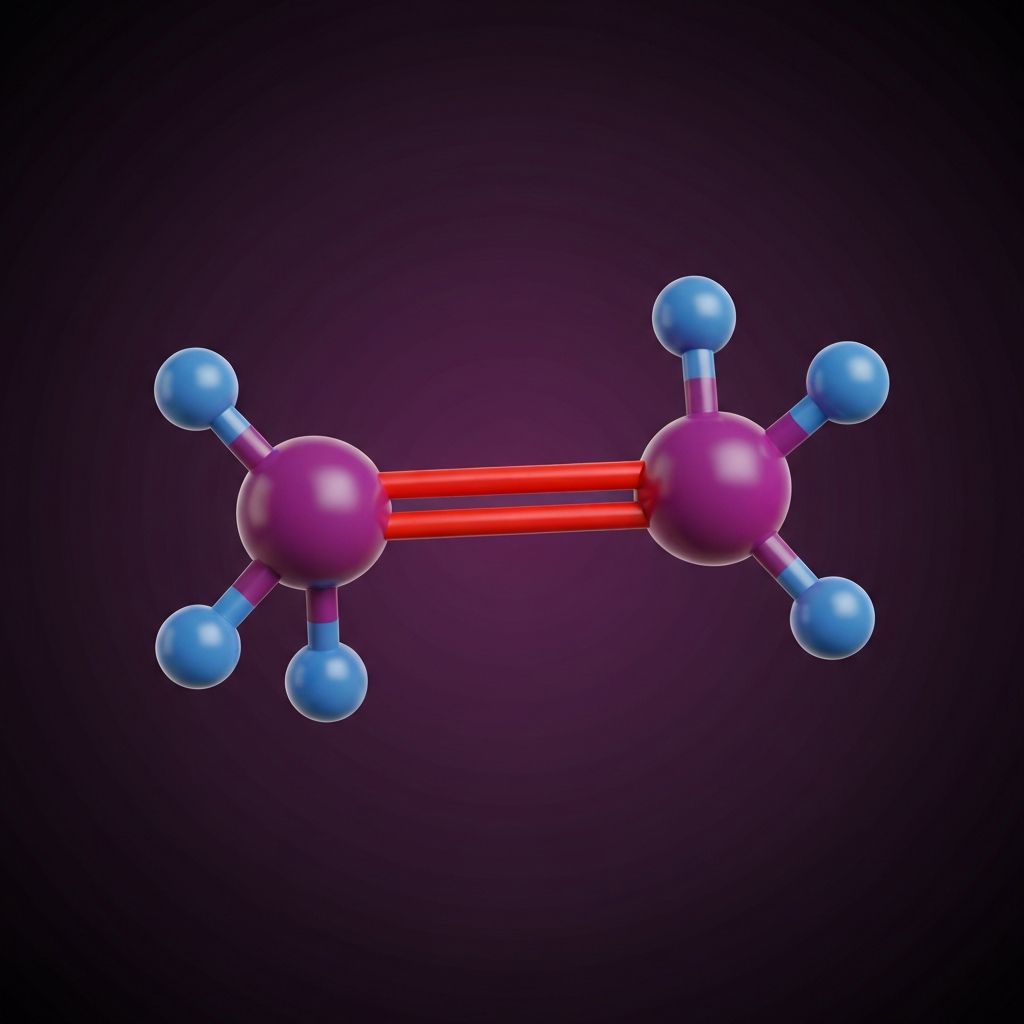
The C=C Double Bond
Reactive center of alkene chemistry
Alkenes are unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C). Unlike alkanes which are saturated (maximum hydrogen atoms), alkenes have two fewer hydrogen atoms because the double bond uses electrons that would otherwise bond to hydrogen.
The general formula for alkenes is CₙH₂ₙ. Compare this to alkanes (CₙH₂ₙ₊₂) – for the same number of carbons, alkenes always have 2 fewer hydrogen atoms. The first four alkenes are ethene (C₂H₄), propene (C₃H₆), but-1-ene, and but-2-ene (both C₄H₈).
The C=C double bond makes alkenes highly reactive. They undergo addition reactions where atoms add across the double bond, breaking it to form a single bond. Three key addition reactions are: hydrogenation (adding H₂ to form alkanes), halogenation (adding Br₂ or Cl₂), and hydration (adding H₂O to form alcohols).
The bromine water test distinguishes alkenes from alkanes: alkenes decolorize orange bromine water (positive test for unsaturation), while alkanes show no color change. Alkenes are important industrial feedstocks used to make polymers (like polyethylene), alcohols (like ethanol), and many other chemicals.
Ethene
Simplest alkene, used to make polyethylene
Molecular Formula
C₂H₄
Structural Formula
CH₂=CH₂
Carbons
2
General Formula: CₙH₂ₙ (alkenes have 2 fewer H atoms than alkanes CₙH₂ₙ₊₂)
Example 1: Drawing Displayed Formula
Question: Draw the displayed formula for propene (C₃H₆).
Answer: Propene has 3 carbon atoms with a double bond between C1 and C2.
CH₃ — CH = CH₂
The displayed formula shows all bonds: H₃C—CH=CH₂ with the C=C double bond clearly shown.
Example 2: Predicting Reaction Products
Question: Write the equation for ethene reacting with bromine.
Answer: This is an addition reaction. Bromine adds across the double bond.
C₂H₄ + Br₂ → C₂H₄Br₂
Each carbon gains one Br atom. Product is 1,2-dibromoethane (colorless liquid).
Example 3: Explaining the Bromine Test
Question: Explain why bromine water decolorizes with alkenes but not with alkanes.
Answer: Alkenes contain a C=C double bond which can undergo addition reactions. Bromine molecules add across the double bond, forming a colorless dibromo compound. The orange Br₂ is consumed in the reaction, so the solution becomes colorless.
Alkanes only have single C-C bonds and are saturated. They cannot undergo addition reactions at room temperature, so the bromine remains unreacted and the solution stays orange.
What is an alkene?
Click to reveal • Card 1 of 14
What is the general formula for alkenes?