Thermal Energy Transfer
Understanding conduction, convection, and radiation
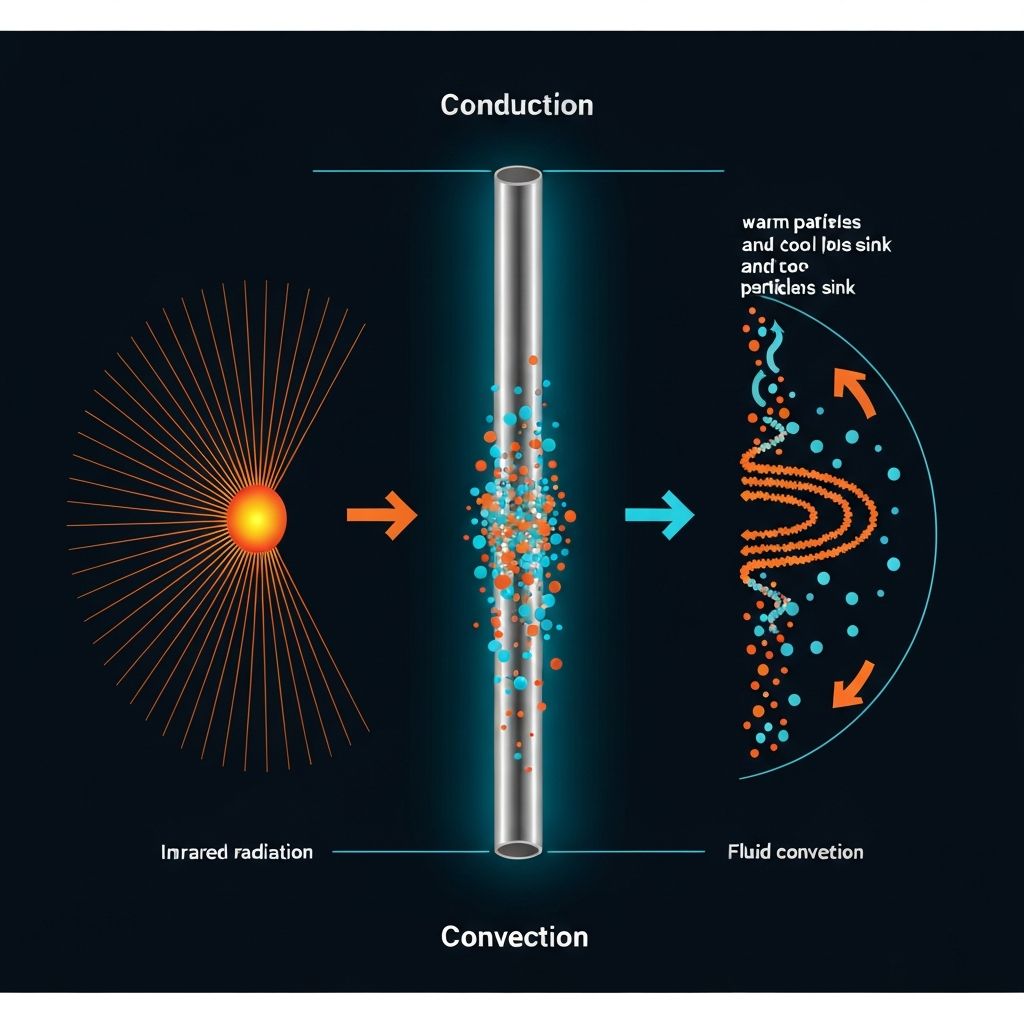
Three ways heat moves: particles vibrating, fluids circulating, and electromagnetic waves
Thermal energy always flows from hot regions to cold regions. There are three methods of heat transfer: conduction (through solids via particle vibrations), convection (in fluids via particle movement), and radiation (via infrared electromagnetic waves, requiring no medium).
Conduction in Solids
When one end of a solid is heated, particles gain kinetic energy and vibrate faster. These vibrations transfer to neighboring particles through collisions, passing energy along without the particles themselves moving position. Metals are excellent conductors because they have free electronsthat move rapidly through the material, transferring energy much faster than vibrations alone. Insulators like wood and plastic lack free electrons, making them poor conductors.
Convection in Fluids
Convection occurs in liquids and gases where particles can move freely. When fluid is heated, particles gain kinetic energy, move further apart, and the fluid becomes less dense. This less-dense warm fluid rises, while cooler, denser fluid sinks to replace it, creating a continuous convection current. This is why radiators are placed low in rooms and freezers have cooling elements at the top.
Radiation Through Space
Unlike conduction and convection, radiation requires no particles at all. All objects emit infrared radiation, with hotter objects emitting more. Dark, matt surfaces are better emitters and absorbers; light, shiny surfaces reflect radiation. This is why space blankets are shiny and why houses with dark roofs absorb more heat in summer.
Reducing Heat Loss
Home insulation uses different methods to block each transfer type: double glazingtraps air (poor conductor) in a gap too thin for convection; cavity wall insulationfills wall gaps with foam containing trapped air pockets; loft insulation uses fiberglass to trap air; and reflective foil behind radiators reduces radiation loss. A vacuum flask combines all three methods: vacuum prevents conduction and convection, while shiny surfaces reduce radiation.
Conduction transfers heat through particle vibrations. In Copper (Metal)(conductivity: 401 W/m·K), particles vibrate faster when heated and pass energy to neighboring particles. Metals conduct best due to free electrons.
| Method | Requires Particles? | Works in Vacuum? | How it Works |
|---|---|---|---|
| Conduction | Yes | No | Particle vibrations transfer energy through collisions |
| Convection | Yes (fluids only) | No | Hot fluid rises, cool fluid sinks - circulation |
| Radiation | No | Yes | Infrared electromagnetic waves |
Example 1: Why metal feels colder than wood
Question: A metal door handle and wooden door are both at room temperature (20°C). Explain why the metal handle feels colder when you touch it.
Answer:
Both objects are at the same temperature (20°C), which is lower than body temperature (37°C).
Metal is a much better thermal conductor than wood due to its free electrons.
When you touch the metal, heat conducts away from your hand much faster than with wood.
This rapid heat loss from your skin makes the metal feel colder, even though both are the same temperature.
Example 2: Convection current in a room
Question: Describe how a radiator warms a room using convection.
Answer:
- The radiator heats the air directly next to it by conduction and radiation
- This heated air expands, becomes less dense, and rises upward
- Cooler, denser air from elsewhere in the room moves in to replace the rising warm air
- The warm air cools as it reaches the ceiling and other parts of the room
- The cooled air becomes denser and sinks back down toward the floor
- This creates a continuous convection current that circulates warm air throughout the room
Example 3: Vacuum flask design
Question: Explain how a vacuum flask minimizes heat transfer by all three methods.
Answer:
- Conduction: The vacuum between the double walls contains no particles, so heat cannot conduct across the gap. The thin glass walls also minimize conduction paths.
- Convection: A vacuum contains no fluid particles, so convection currents cannot form between the walls.
- Radiation: The inner surfaces are coated with shiny silver, which reflects infrared radiation back into the flask (or outward if keeping contents cold), minimizing radiation loss.
- Additional: The stopper/lid is made of insulating material to reduce heat loss through the top.
Conduction
Click card to flip
Which method of heat transfer does NOT require particles?